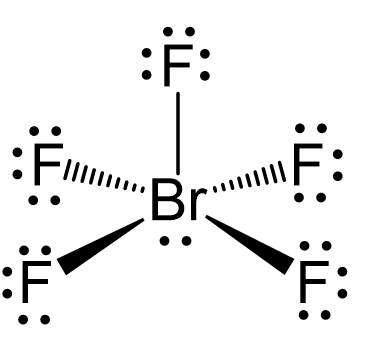
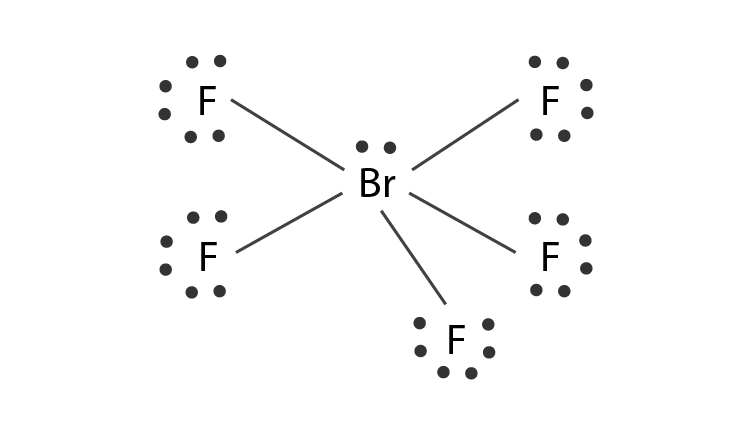
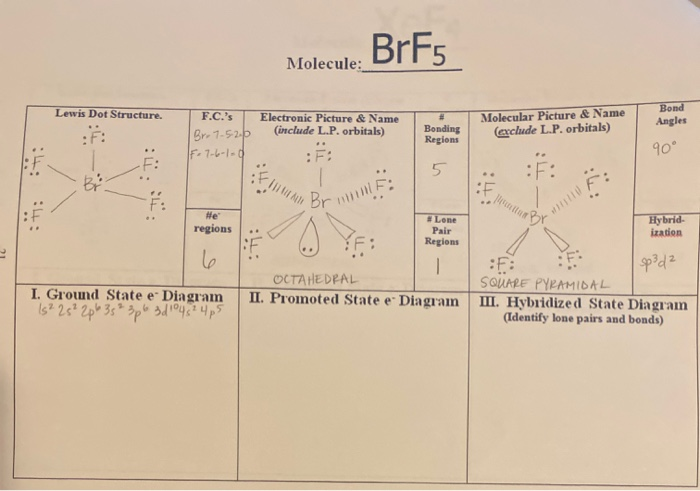
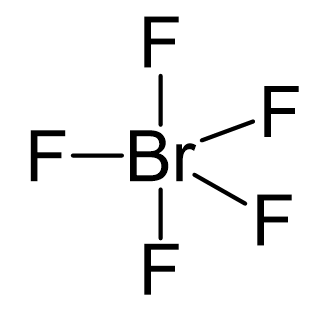
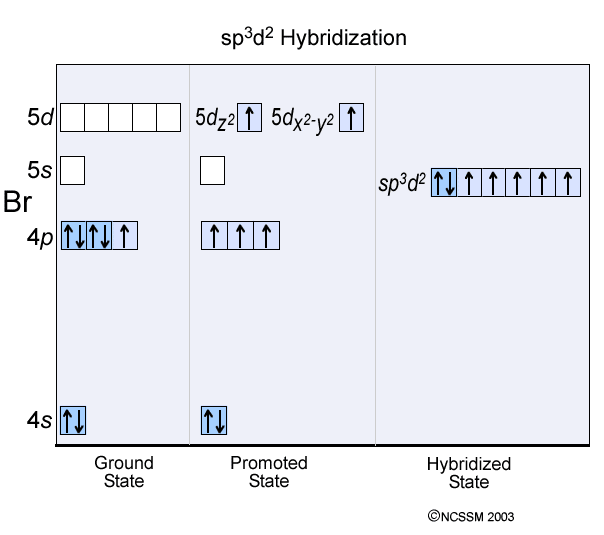
12. Which of the following compounds exhibit d 2sp 3 hybridization? (Select all that apply.) BrF5 ClF5 KrCl4 XeCl2 PCl5 13. Draw the Lewis structure for IF3 and answer the following question.
SOLVED: CHEMWORK: Give the expected hybridization for the central atom for each of the following molecules or ions: Molecule or ion: IF3, CIF5, SeCl6, KrF4, BrF5 Hybridization: Submit Submit Answer Try Another