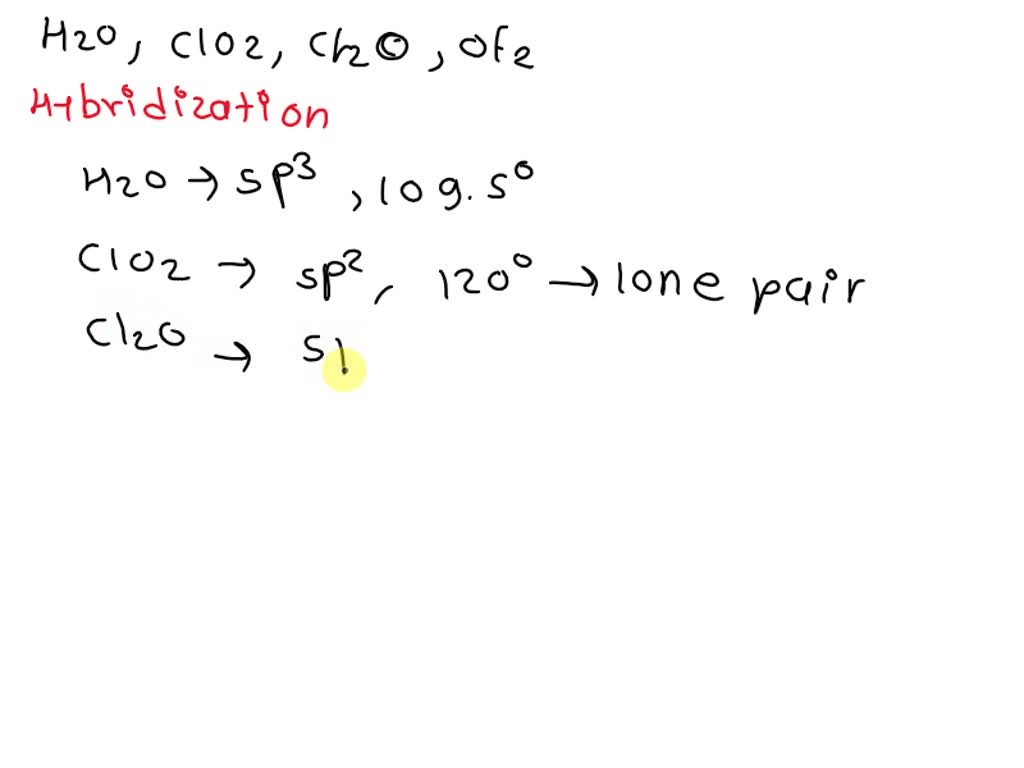Hybridization of Chlorine in Hypochlorite ClO-, Chlorite ClO2-, Chlorate ClO3-, Perchlorate ClO4- - YouTube

The type of hybrid orbitals used by the chlorine atom in \( \mathrm{ClO}_{2}^{-} \)is: (A) \( s ... - YouTube

Which of the following is incorrect statement regarding compound Cl 2 O 6 ?A. In covalent gaseous state, each Cl atom has a hybridisation of sp 3 in Cl 2 O 6B.
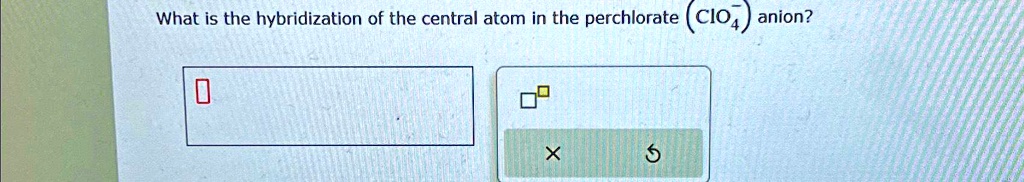
SOLVED: What is the hybridization of the central atom in the perchlorate ( ClO(4)^(-))anion? ◻◻ What is the hybridization of the central atom in the perchlorate (CIO4) anion? 5

Hybridization of Chlorine in Hypochlorite ClO-, Chlorite ClO2-, Chlorate ClO3-, Perchlorate ClO4- - YouTube

The hybridization of Cl atom in ClO_(4)^(-) and ClO_(3)^(-) is | CLASS 12 | NONE | CHEMISTRY | D... - YouTube

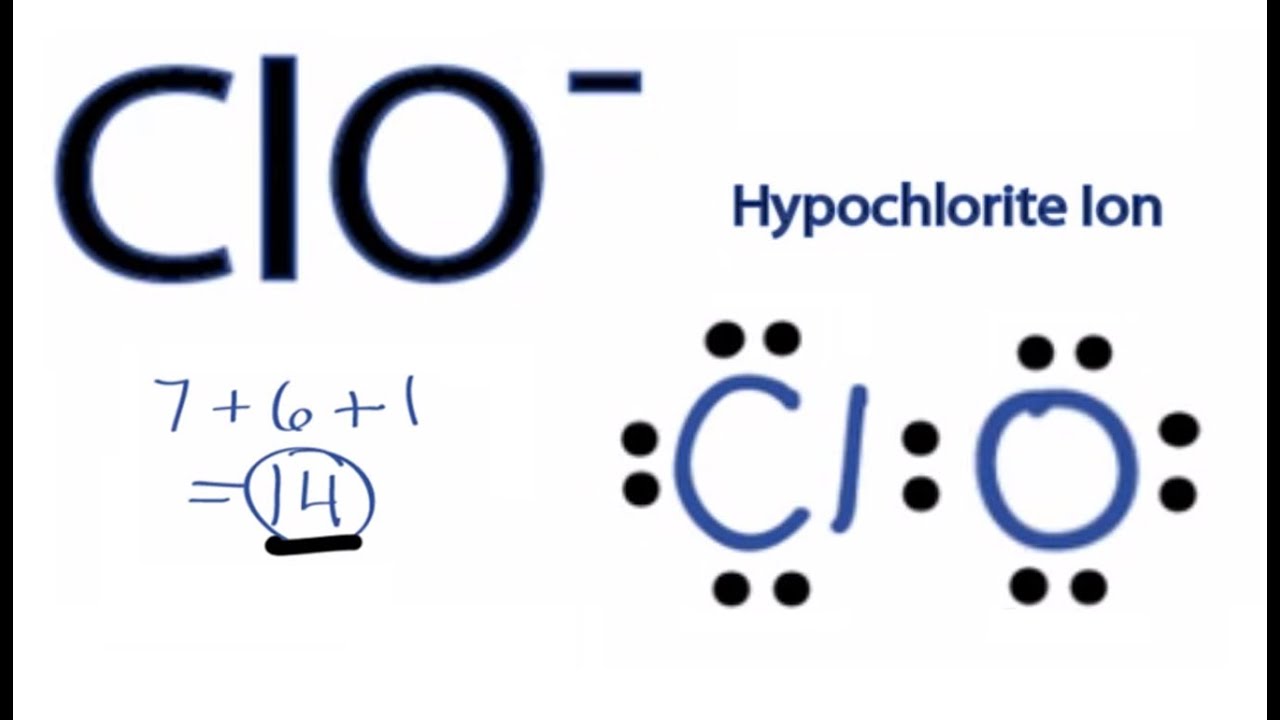
What is the steric number of ClO- ? Specify and draw ClO- electron pair geometry. What is ClO- hybridiztion | Homework.Study.com