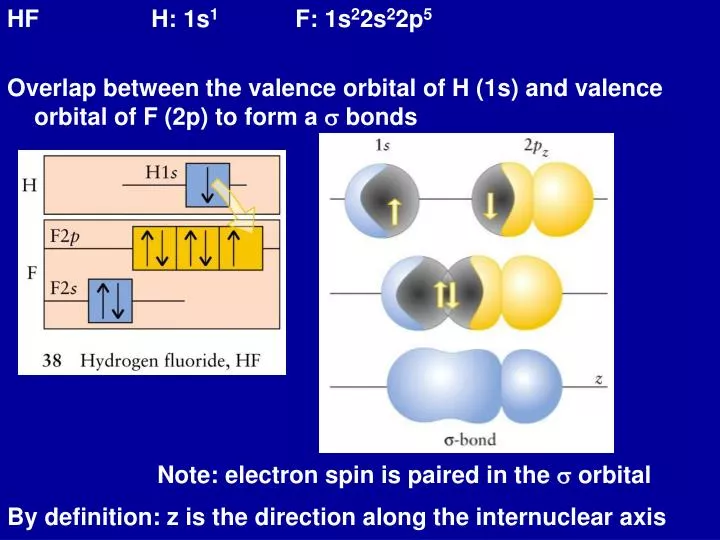
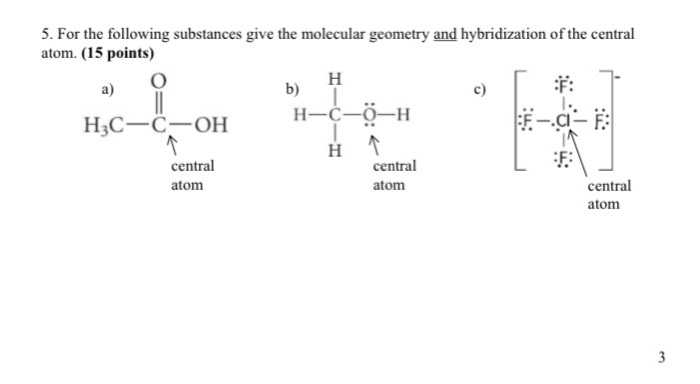
Plots of the hybridization coefficient M versus hf product for the S 4... | Download Scientific Diagram

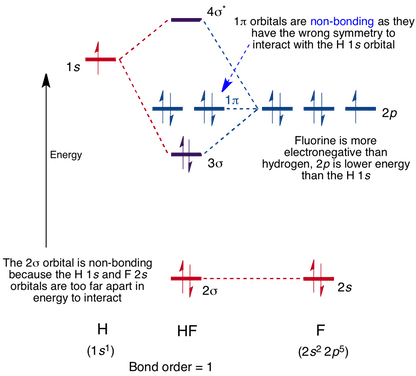
8.4 | Use valence bond theory to explain the bonding in F2, HF, and ClBr. Sketch the overlap of the - YouTube
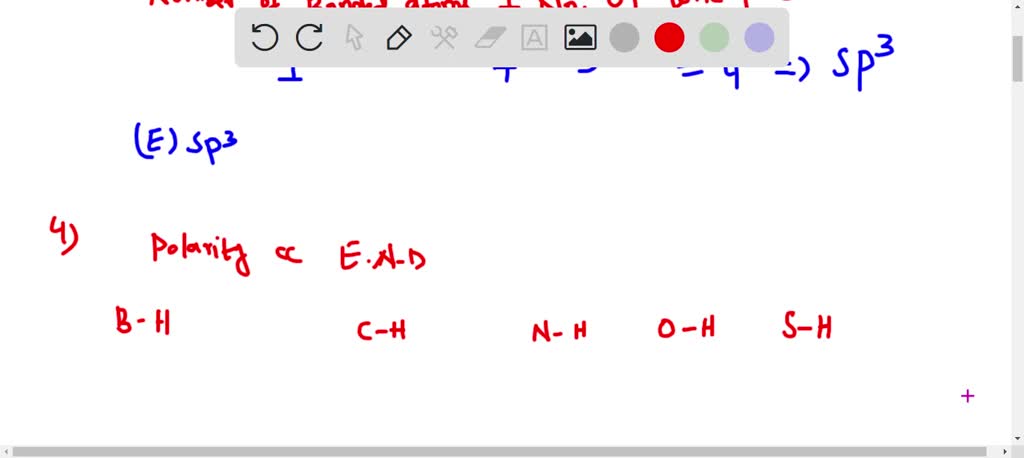
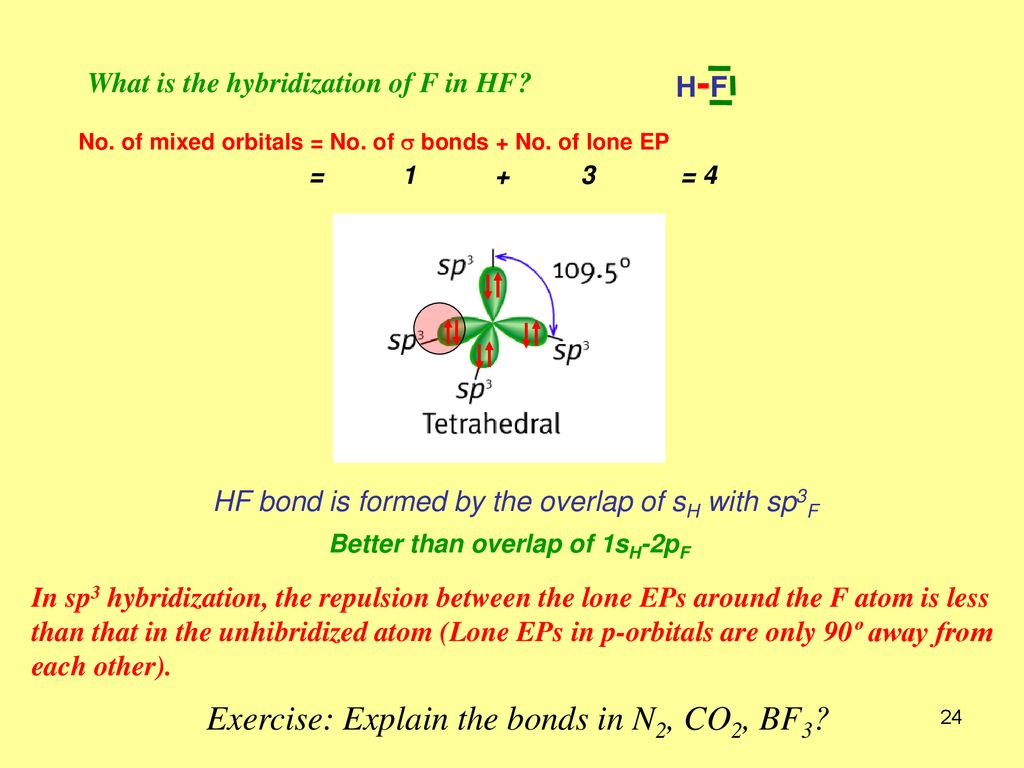
SOLVED: Whar IS the hybridization of fluorine in HF? C sp Which of these bonds 1s the most polar? B-H C-H C; N-I O-H E S-H

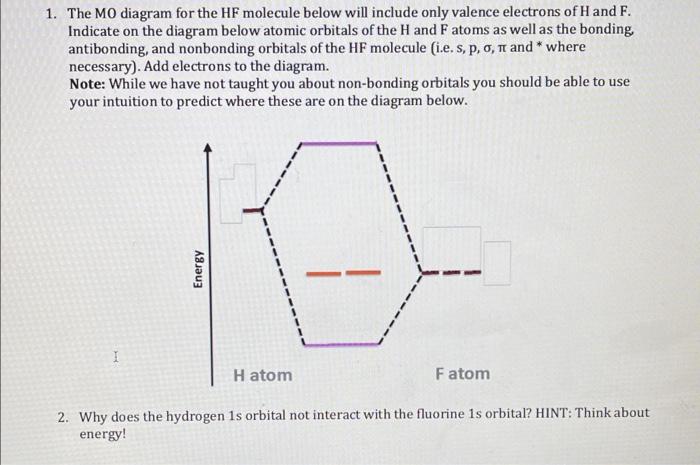
SOLVED: The molecule hydrogen fluoride (HF) contains a polar bond H - F, where fluorine is more electronegative than hydrogen. Which is the appropriate representation of the H - F bond?