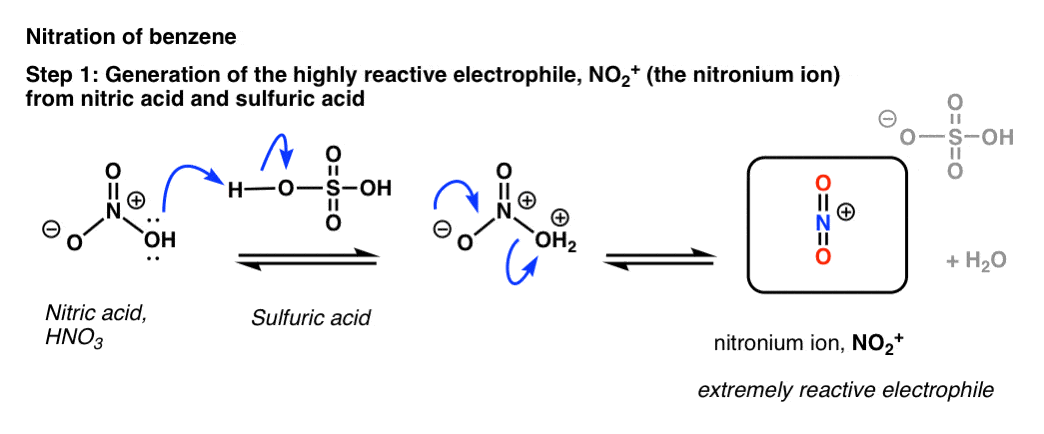
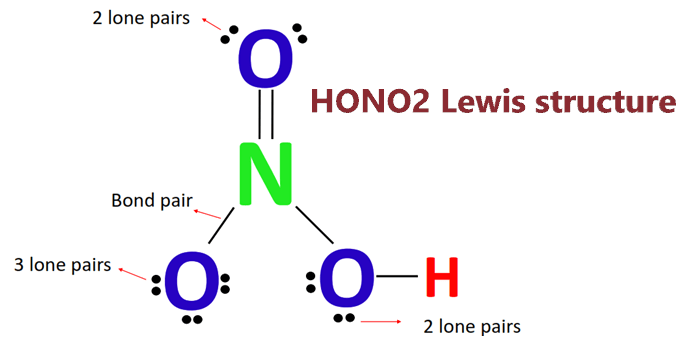
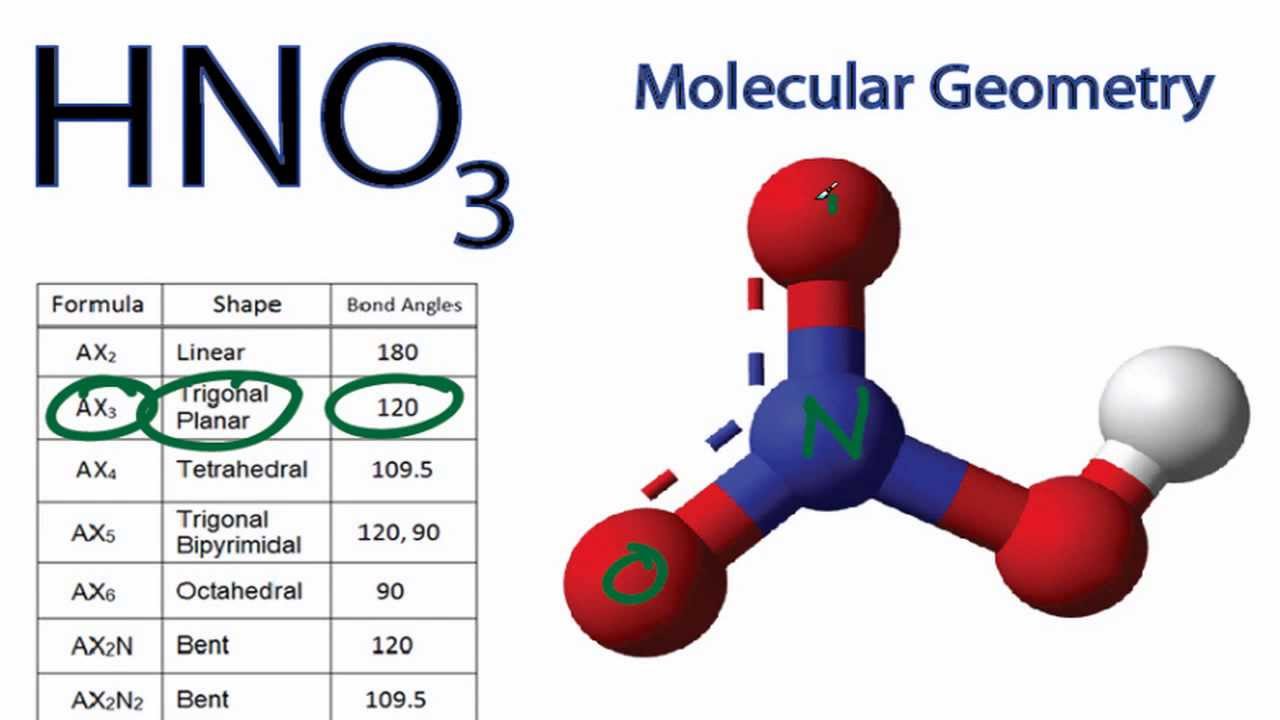
In HNO3 why does the nitrogen atom give electrons to the third oxygen when it already satisfied its valency with two oxygens one with a single bond and one with a double

Nitric acid (HNO3) Lewis structure, molecular geometry, hybridization, polar or nonpolar | Molecular geometry, Molecular, Electron configuration