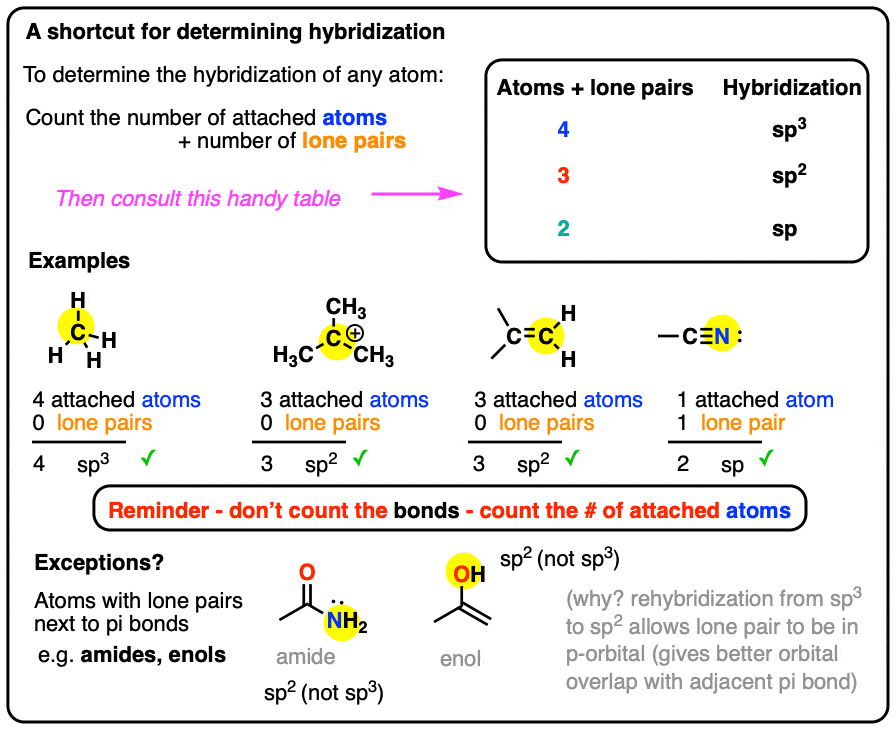
Practical four-electron zinc-iodine aqueous batteries enabled by orbital hybridization induced adsorption-catalysis - ScienceDirect

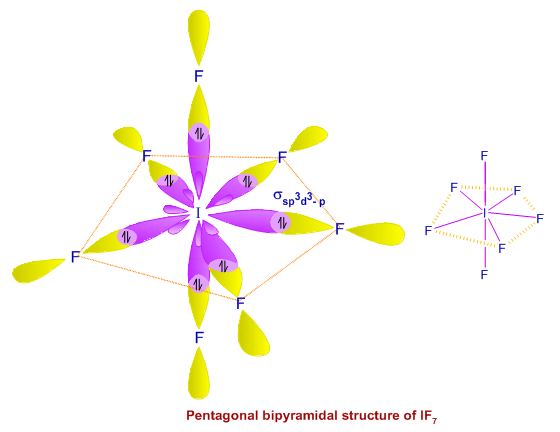
Iodine pentafluoride IF5: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –

Structure of IF_4^+and hybridization of iodine in this structure is, respectively, (A) linear, s ... - YouTube

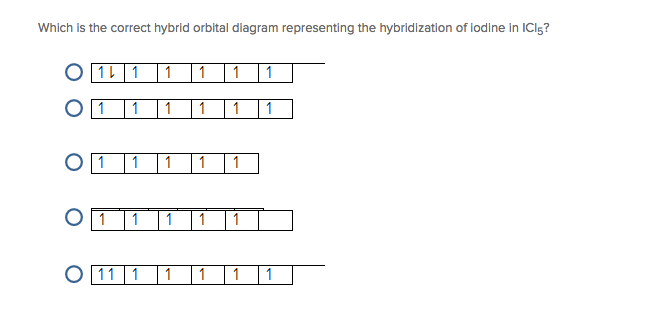
Write down the Lewis structure of ICl3. Provide the electron pair geometry, molecular geometry, and hybridization of iodine. | Homework.Study.com
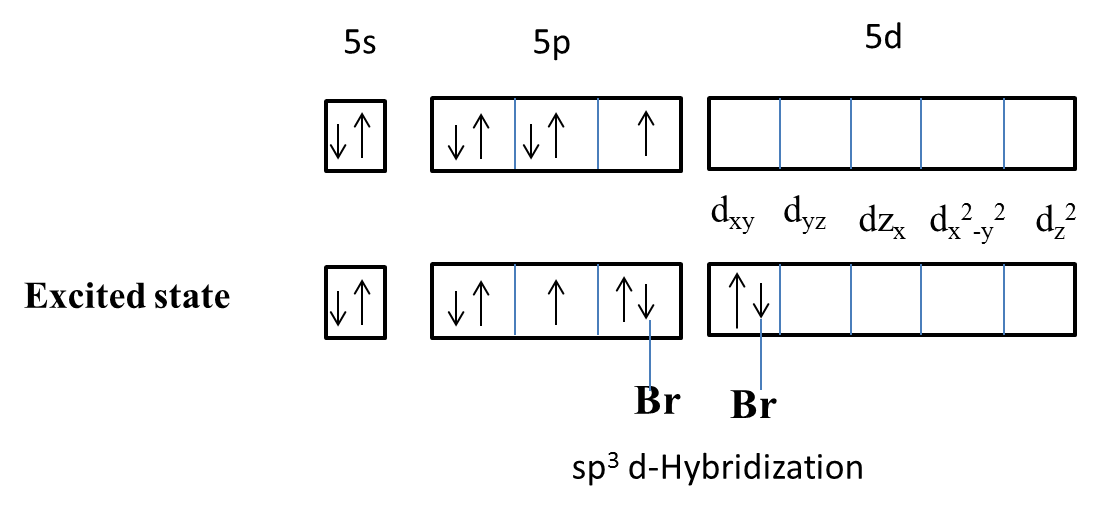
The structure of $IBr_{2}^{-}$ involves hybridization of the type:(A) $s{{p}^{3}}$(B) $s{{p}^{3}}d$(C) $s{{p}^{3}}{{d}^{2}}$(D) None of these