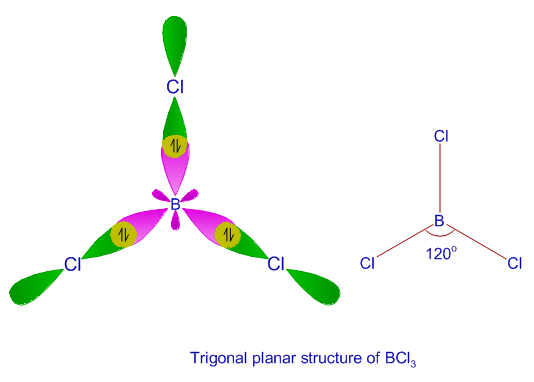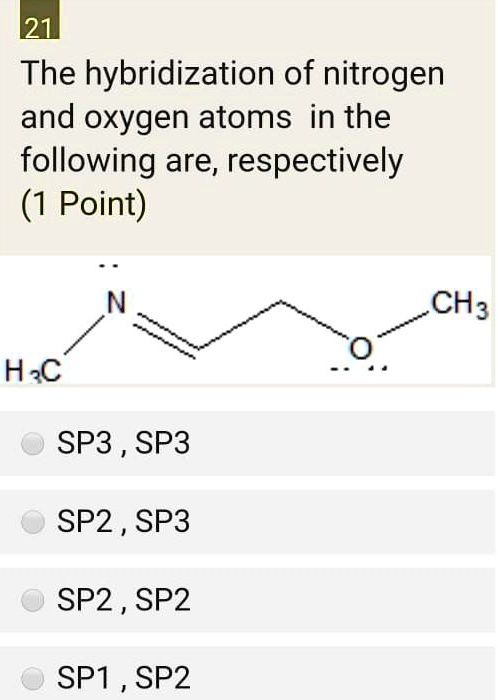
SOLVED: The hybridization of nitrogen and oxygen atoms in the following are; respectively (1 Point) N CH4 O H3C SP3 SP3 SP2 SP3 SP2 SP2 SP1 SP2

sp2 to sp3 Hybridization Transformation in Ionic Crystals under Unprecedentedly Low Pressure - Jiang - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Difference Between sp sp2 and sp3 Hybridization | Definition, Calculation of S and P Characteristics, Features and Differences
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
The ratio of the number of sp, sp2, sp3 orbitals in the compound is CH3 CH=C=CH CC CH3 A)1:1:1 B)2:2:1 C)3:2:1 D)3:3:4
Catalysis by hybrid sp2/sp3 nanodiamonds and their role in the design of advanced nanocarbon materials