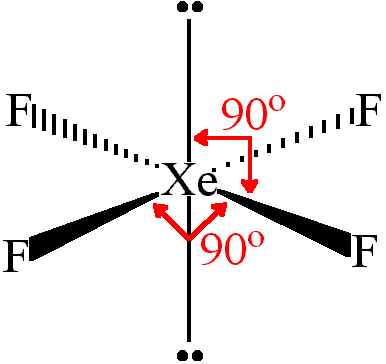
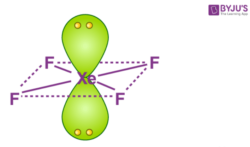
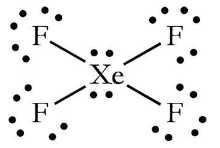
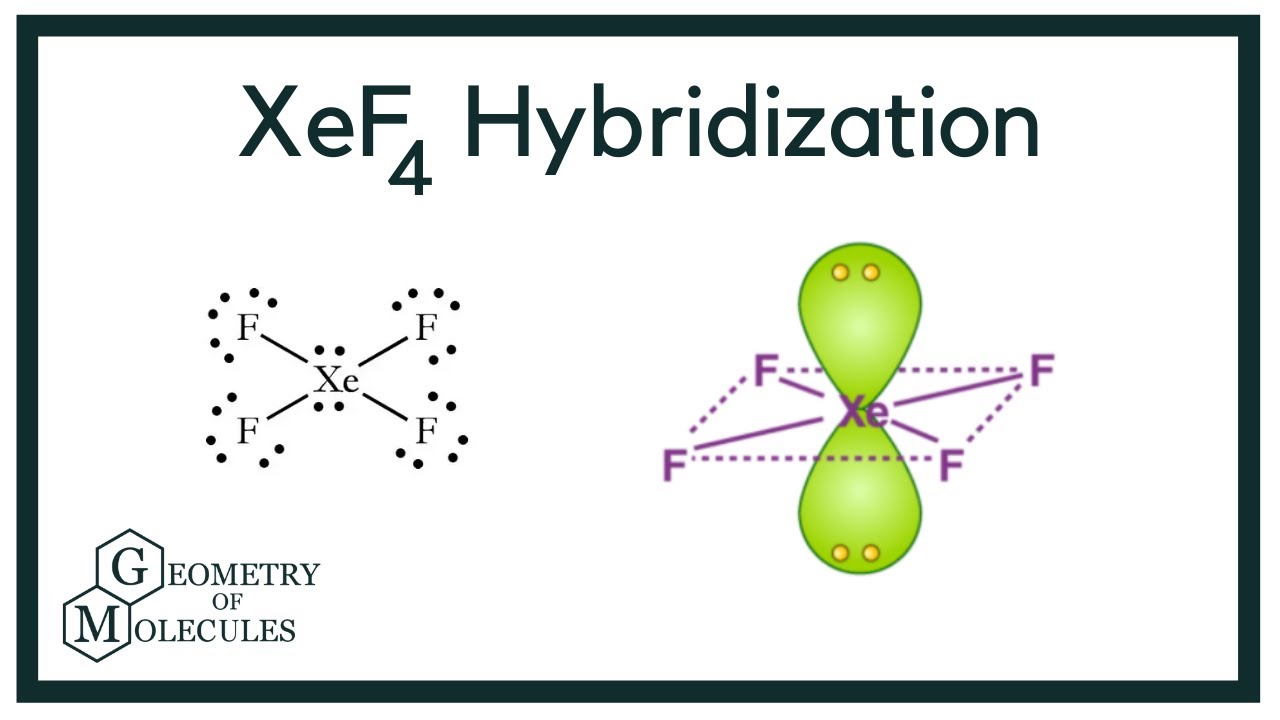
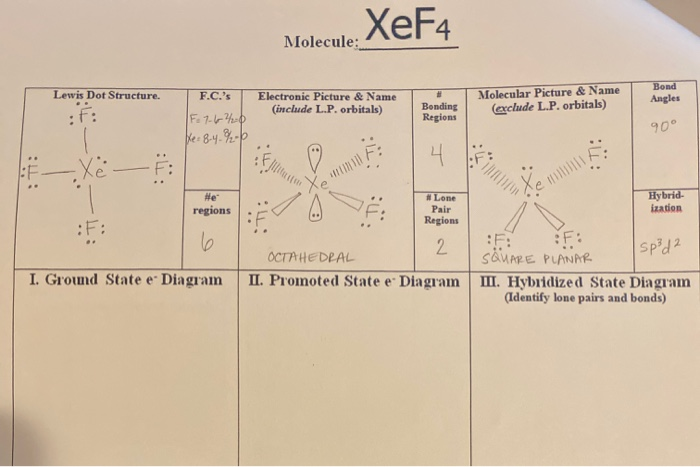
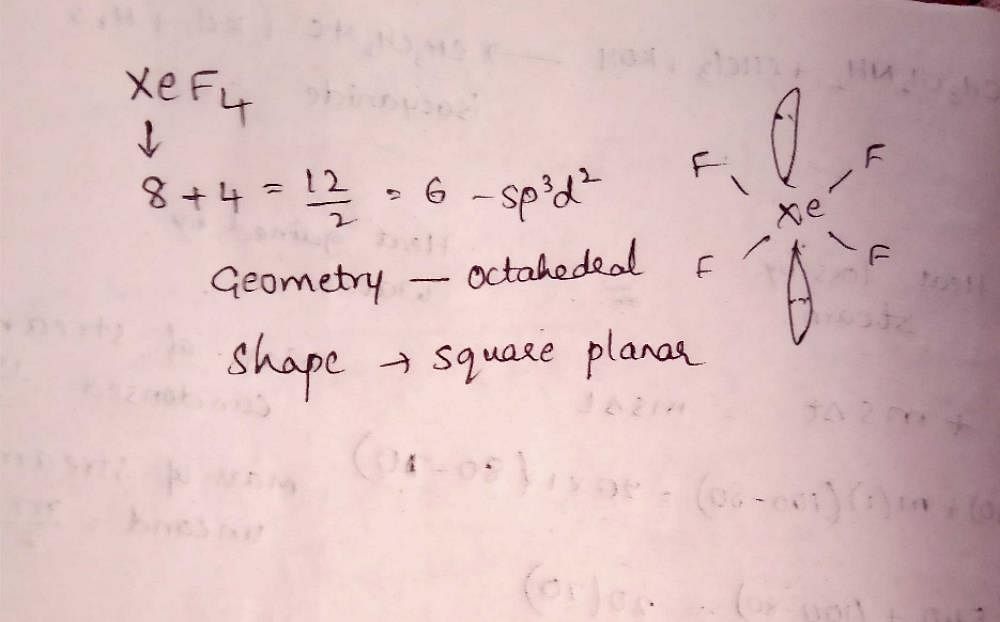Hybridization and shape of mathrm{X}mathrm{e}mathrm{F}_{4} is: mathrm{s}mathrm{p}^{3}mathrm{d}, trigonal bipyramidal mathrm{s}mathrm{p}^{3}, tetrahedral mathrm{s}mathrm{p}^{3}mathrm{d}^{2}, square planar mathrm{s}mathrm{p}^{3}mathrm{d}^{2}, hexagonal

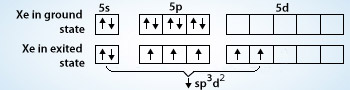
Chemistry Help on Instagram: "How is Xe atom hybridized in XeF4? Xenon tetrafluoride (XeF₄) is a compound that exhibits sp³d² hybridization. Hybridization is a concept in chemistry that describes the mixing of

Hybridization of Xe in XeF4 is ........ and in XeF2 is ....... a. sp3, sp3d b. sp3d2, sp3d2 c. sp3d, sp3d2 d. sp3d2, sp3d e. sp3, sp3d2 | Homework.Study.com




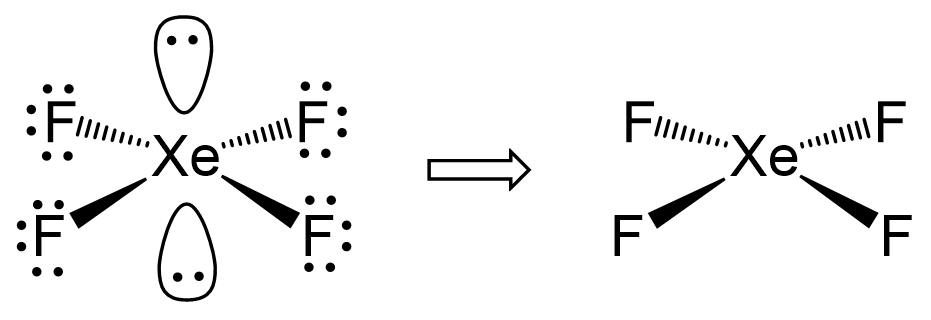
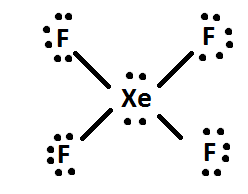


![ANSWERED] A. What is the hybridization of the central atom in XeF4 - Kunduz ANSWERED] A. What is the hybridization of the central atom in XeF4 - Kunduz](https://media.kunduz.com/media/sug-question/raw/52219992-1659248456.8030424.jpeg)